If the solution are equally concentrated the amount of rototation caused by the two isomers is exactly the same , but in opposite directions. When optically active substance are made in the lab, they often occurs as a 50/50 mixture of the two enantiopmers. This is known as a racemic mixture or racemate. It has no effect on plane polarised light.
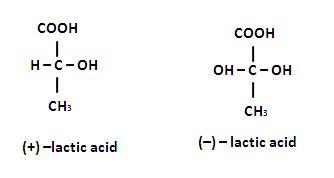
Which of the following have the same rates of reaction ?
(a) Reaction of (+) and (-) enantiomers with a chiral reagent.
(b) Reaction of diastereomers with a chiral reagent .
(c) Reaction of (+) and (-) enantiomers with an achiral reagent.
(d) Reaction of diastereomers with an achiral reagent.
