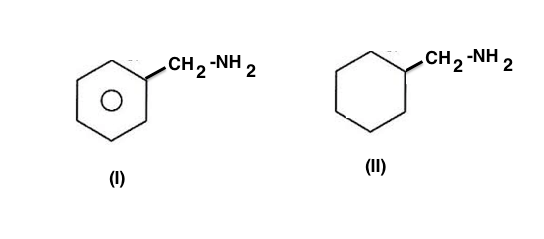
$ \begin{array}{1 1}(a)\;\text{(I) > (II) because it is an aromatic amine}\\(b)\;\text{(II) > (I) because it is an aliphatic amine}\\(c)\;\text{(I) < (II) because of difference in the nature of }\beta-carbon\\(d)\;\text{Both are equally basic because both are }1^o\text {amines}\end{array}$
